
Essential protection from long-term complications
For a long time, soft tissue management was seen as purely esthetic. But around implants both keratinized tissue and sufficiently thick soft tissue have protective functions.
In our practice, soft tissue management - in the form of flap advancement, rolled flap, connective tissue or collagen matrix grafting - is part of the treatment in approximately 80 percent of our cases.
What soft tissue conditions are beneficial?
In terms of the quality of the surrounding soft tissue, augmenting an approximately 2 mm band of attached keratinized mucosa should be the goal to establish stable, long-term conditions. One review concluded that a lack of adequate keratinized mucosa is associated with more plaque accumulation, tissue inflammation, mucosal recession and attachment loss.1
As to the quantity (thickness and height of the peri-implant soft tissue cover), studies show that thicker peri-implant soft tissue leads to less bone resorption. Thus, Linkevicius et al. were able to show that
2 mm thick peri-implant soft tissue induces significantly less crestal bone loss than a soft tissue thickness of less than 2 mm.2 This phenomenon may be explained by the necessary establishment of an apical biological width with thin marginal mucosa and two-part implants. The group was able to show that peri-implant soft tissue augmented with biomaterials behaves in a similar way to "naturally thick” soft tissue.2 It was also determined, using the highest level of evidence (Osteology Consensus Conference), that soft tissue augmentations protect against crestal bone resorption.3 Today it must therefore be concluded that the target for both soft tissue thickness and attached mucosa should be
2 mm (Fig. 1).
Techniques for gaining keratinized mucosa
Free mucosal transplants are the gold standard for augmenting attached keratinized mucosa. Free mucosal transplants, however, have various other disadvantages in addition to painful removal and the increased risk of complications. These include scarring and insufficient adaptation to the surrounding soft tissue. According to a retrospective case control study from our working group, covering extraction sockets with gingiva punch products produces considerably more scars, contractions and color deviations than the collagen matrix Geistlich Mucograft® Seal.4 We consider Geistlich Mucograft® Seal to be preferable as a closure for extraction sockets.
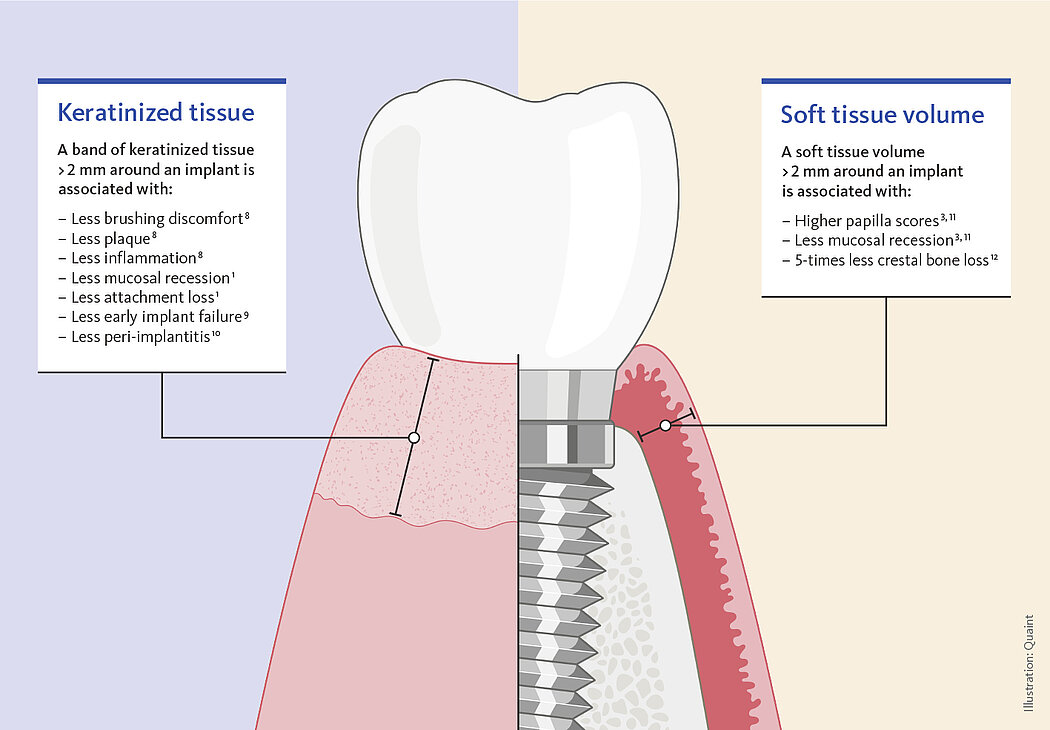
Figure 1. Illustration showing the target amout for the attached mucosa and for the soft tissue.
Techniques for volume thickening
Subepithelial connective tissue transplants are the current gold standard for augmenting the volume of soft tissue around implants. However, clinical studies show that a similar tissue volume can be obtained with a volume-stable xenogeneic collagen matrix (Geistlich Fibro-Gide®).5,6 In this respect this development is interesting for clinicians, as removing subepithelial connective tissue transplants or advancement flap techniques for thickening soft tissue are often very complex and technically sensitive procedures. The introduction of collagenous soft tissue matrices has reduced patients’ surgical burden. These modern methods also allow soft tissue augmentations for increasing tissue thickness in the posterior zone. As an approximate value, the use of the collagen matrix can be assumed to increase soft tissue thickness by 1-1.5 mm. In terms of the 2 mm protective soft tissue thickness requirement, this means that sufficiently thick soft tissue can be achieved in a single soft tissue augmentation.
Points in time
The soft tissue can be improved at different times. A Ridge Preservation can be performed immediately after tooth extraction. Using a porcine collagen matrix (Geistlich Mucograft® Seal) for covering the extraction socket can achieve a better and faster soft tissue closure with only minor scarring.4 Soft tissue can also be thickened at the same time as the implantation. In this context Geistlich Fibro-Gide® is a tested means for improving peri-implant soft tissue thickness prior to uncovering implants. Late soft tissue thickening after the implantation can be more difficult. Scientific data show that secondary soft tissue correction brings little success.7 Accordingly, I prefer proactive soft tissue augmentation to subsequent correction and try to avoid the formation of thin or dehiscent soft tissue around a prosthetically treated implant from the start.
All parameters fulfilled
The clinical case in Figure 2 shows a patient with a thin and poorly attached mucosa. The aim here was to achieve all the targets required for sufficient peri-implant tissue – 2 mm attached mucosa plus
2 mm soft tissue thickness – by augmenting volume and subsequently managing the soft tissue. Geistlich Fibro-Gide® was placed at the time of the implantation to allow primary healing beneath the mucosa. Visibly, Geistlich Fibro-Gide® thickened the soft tissue significantly. The collagen matrix Geistlich Mucograft® was then used with open healing at the time the implants were exposed in order to obtain an augmented band of attached mucosa.
Why this approach? As already alluded to, soft tissue formation after a complication, e.g., implant dehiscence and/or poorly attached mucosa to a prosthetically treated implant, is a difficult and less predictable process. For this reason the above-mentioned soft tissue augmentations prior to prosthetic restoration are key to avoiding middle- and long-term complications.
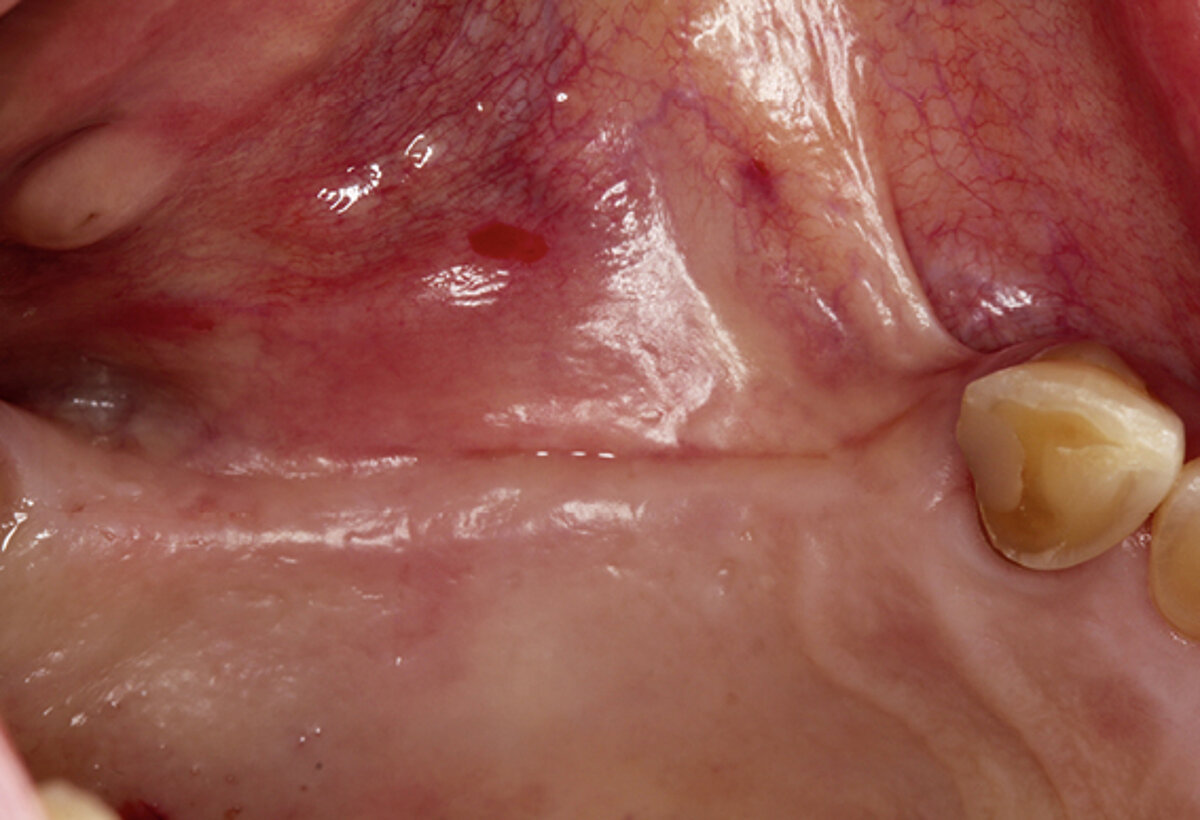
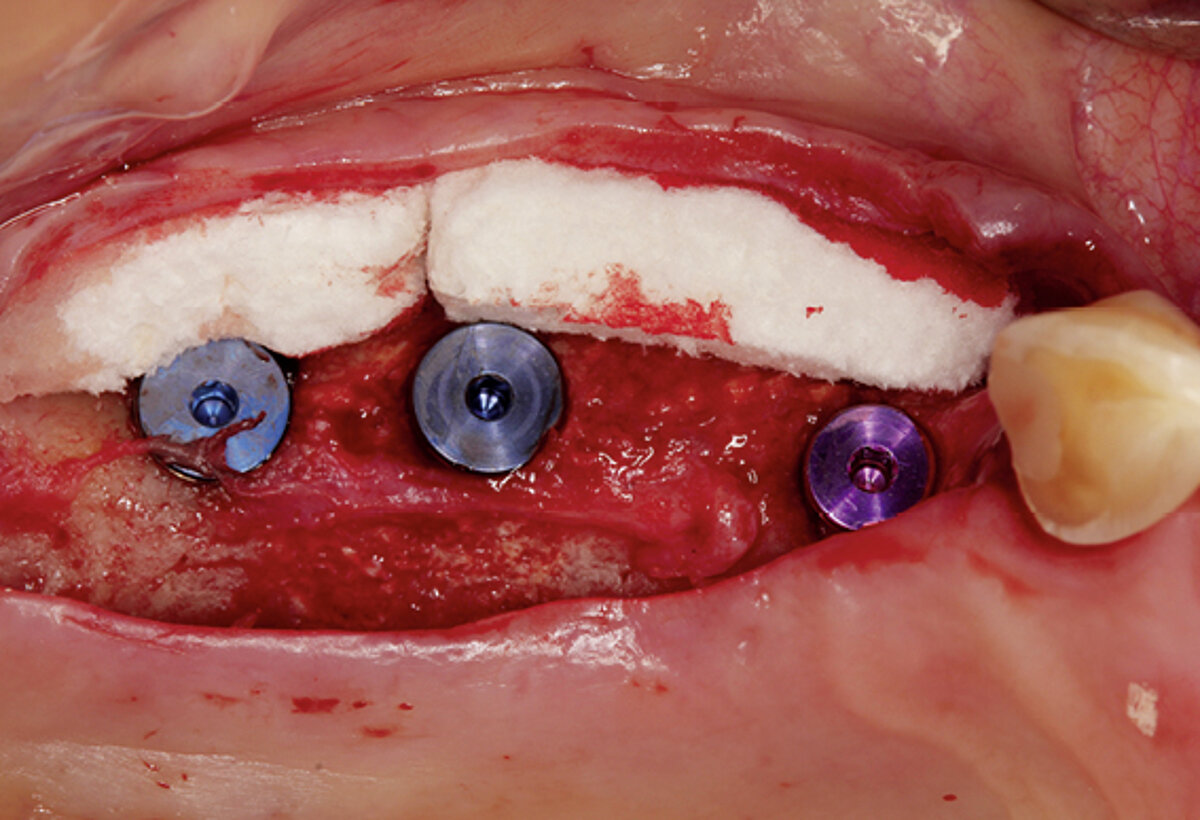
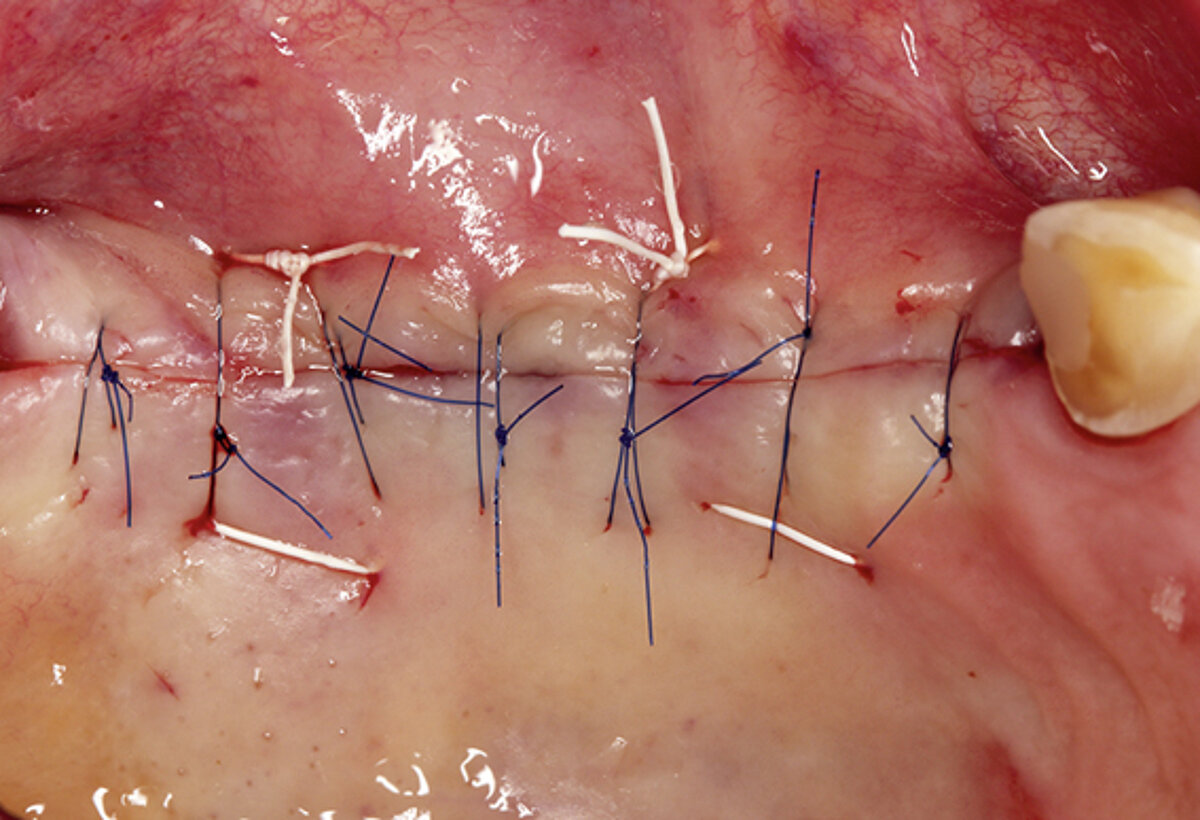
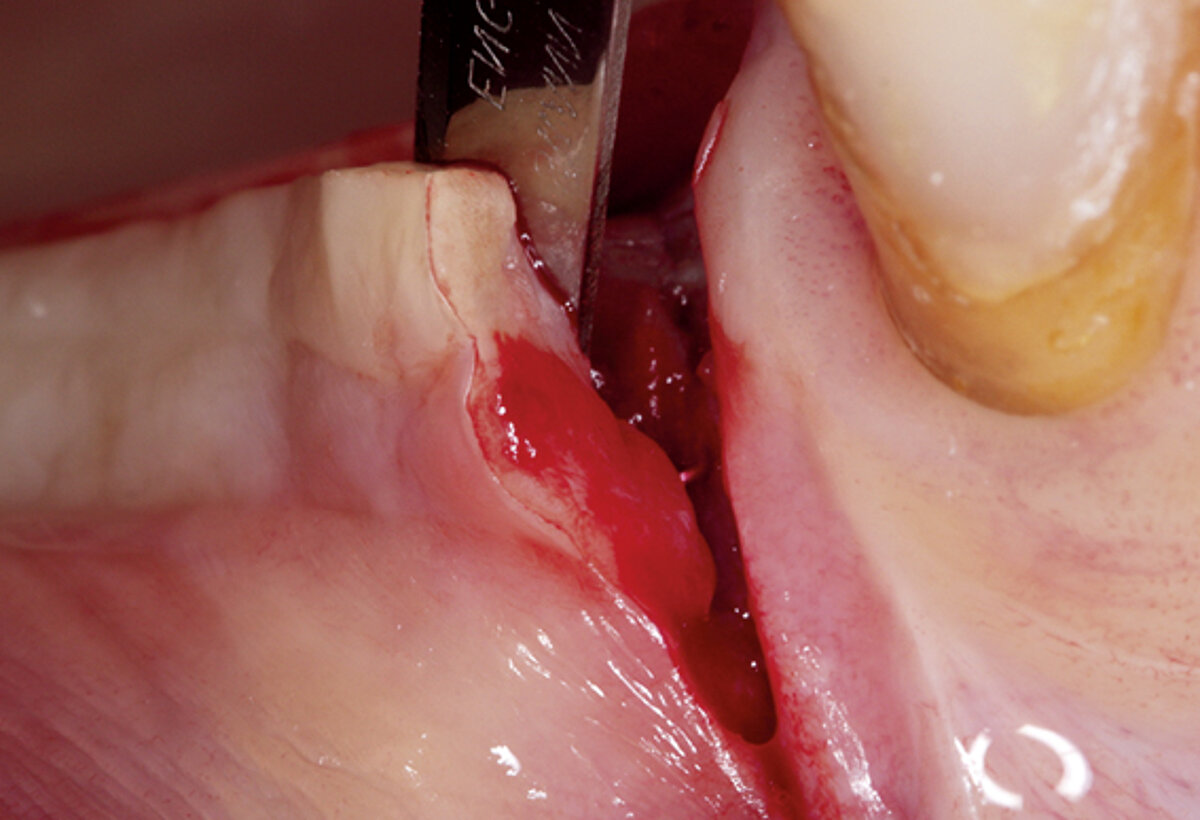
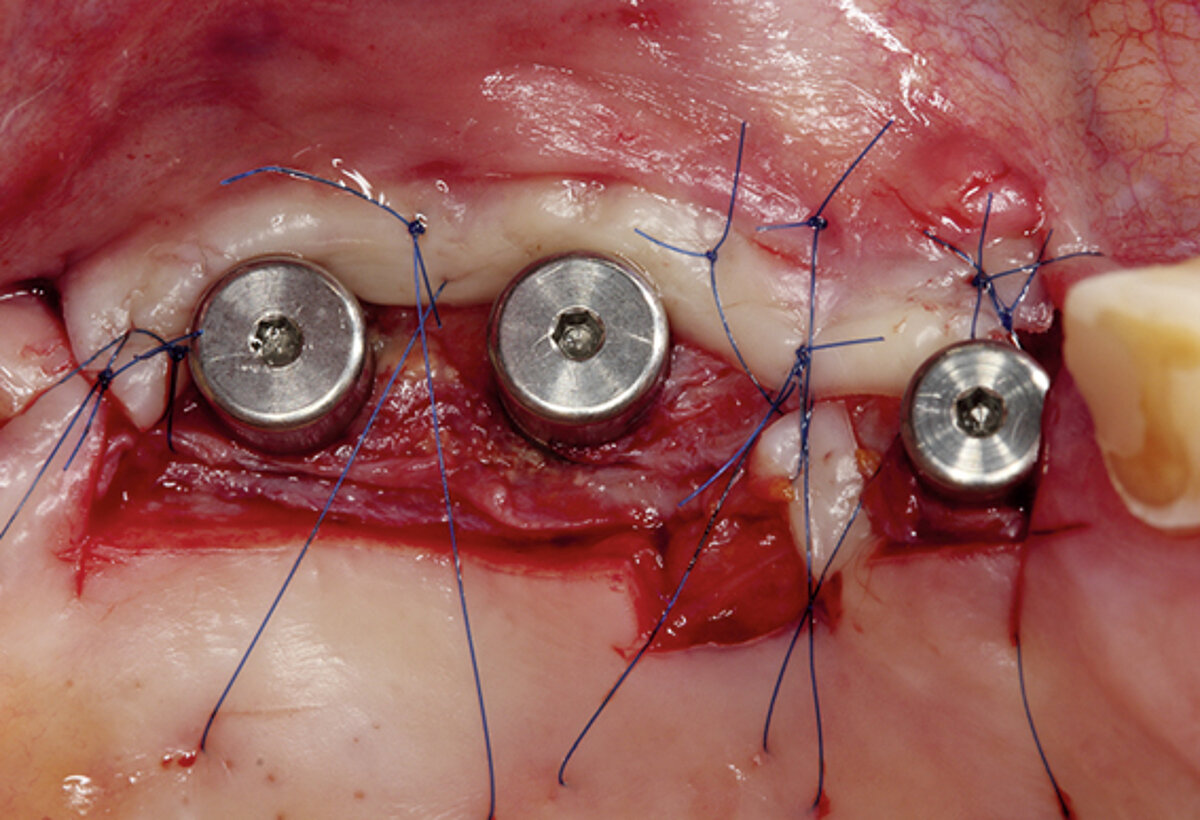
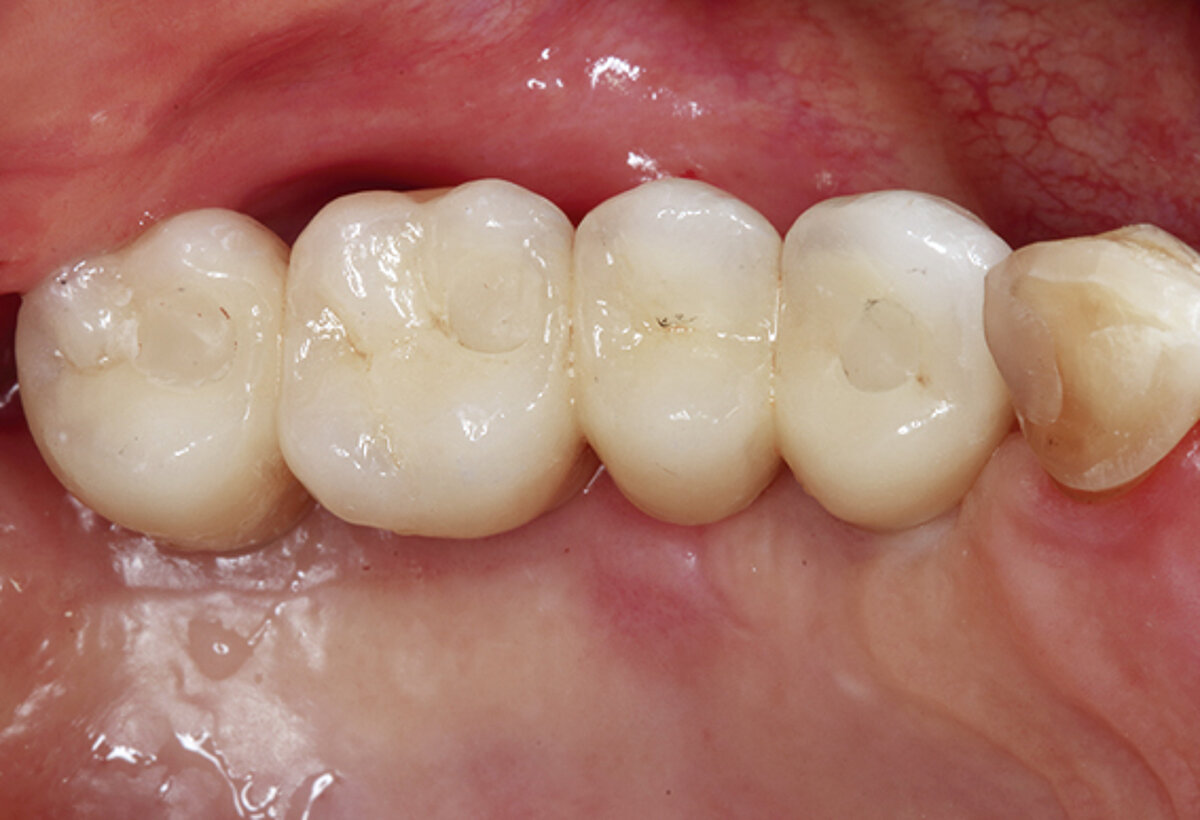
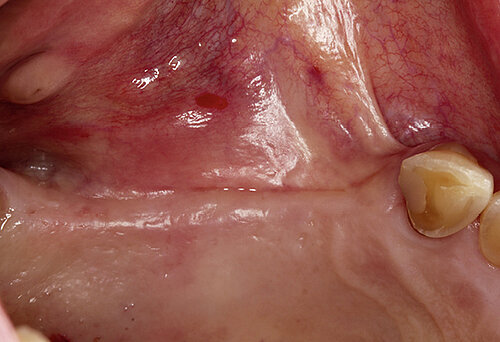
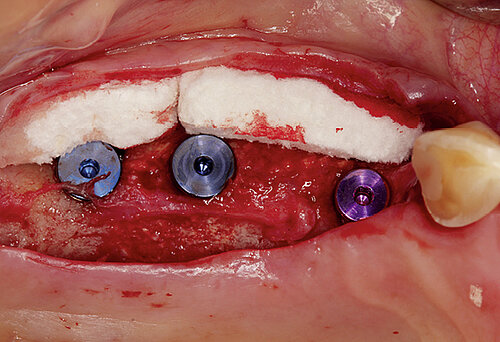
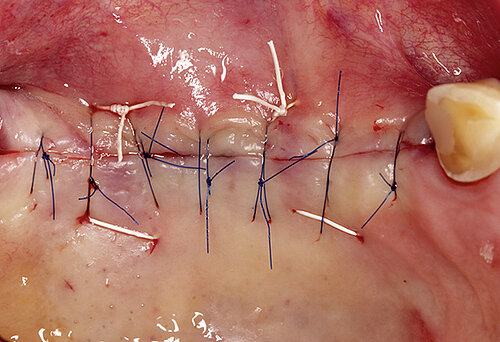
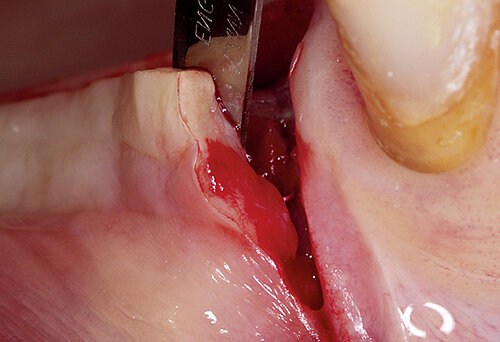
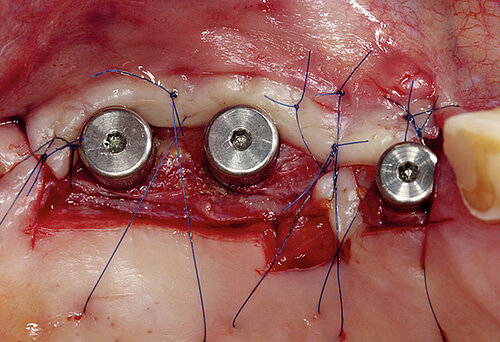
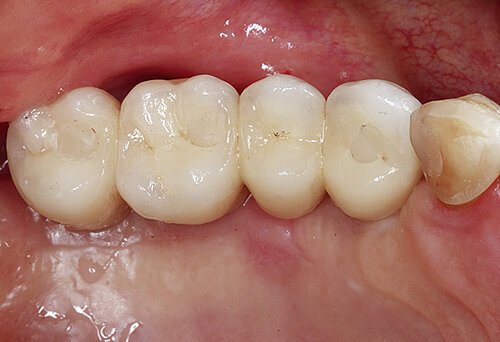
Figure 2. Soft tissue augmentation procedure using Geistlich Fibro-Gide® and Geistlich Mucograft®.
Conclusion for clinical practice
Autologous transplantation of soft tissue may still be the standard technique for improving peri-implant health, but the use of collagen matrices extends the indication and the clinical options for improving soft tissue and ensuring patients a long-term and stable soft tissue peri-implant solution.
References
- Lin GH, et al.: J Periodontol 2013; 84:1755-67.
- Linkevicius T, et al.: Clin Implant Dent Relat Res 2015; 17(6):1228-36.
- Thoma DS, et al.: J Clin Periodontol 2017; 44: 185-194.
- Fickl S, et al.: Int J Periodontics Restorative Dent 2018; 38:e1-e7.
- Thoma DS, et al.: Clin Oral Implants Res 2018; 29 Suppl 15:32-49.
- Zeltner M, et al.: J Clin Periodontol 2017; 44:446-53.
- Burkhardt R, et al.: JClin Oral Implants Res 2008;19(5):451-7.
- Souza AB, et al.: Clin Oral Impl Res 2016; 27:650-55.
- Sarmiento HL, et al.: Int J Periodontics RestorativeDent 2016; 36(5):699-705.
- Baqain ZH, et al.: British Journal of Oral and Maxillofacial Surgery 2012; 50:239-43.
- Thoma DS, et al.: J Clin Periodontol 2016;43(10):874-85.
- Thoma DS, et al.: Clin Oral Implants Res 2014; 41 Suppl 15:S77-91.






